|
Main Topics > Quantum Theory and the Uncertainty Principle > Early Developments in Atomic Theory
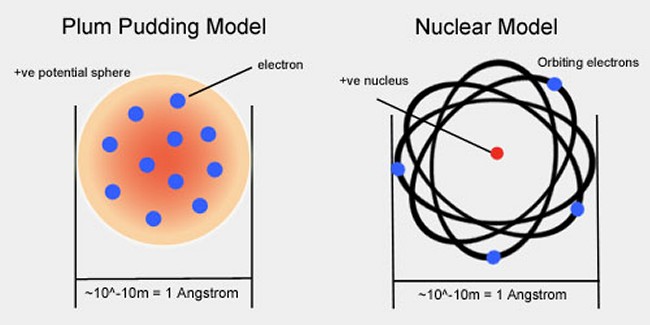
The development of quantum theory was arguably many centuries in the making. As early as the 5th Century B.C., the Greek philosophers Democritus and Leucippus first put forward the idea that everything around us was made of tiny indivisible pieces called atoms scattered in an infinite void. However, finding (even indirect) evidence of such atoms had to wait over two millennia until the work on gases and pressure by the 17th Century English scientist Robert Boyle and, later, the 18th Century Swiss mathematician Daniel Bernoulli, which bolstered the case for the existence of tiny grain-like atoms flying around in empty space. In 1789, the French aristocrat and scientist Antoine Lavoisier identified (albeit slightly incorrectly) 23 elements which he claimed could not be broken down into simpler substances. In the very early 19th Century, the English chemist and physicist John Dalton identified the atomic weights of certain atoms, and first posited the idea that chemical combinations were due to the interaction of atoms of definite and characteristic weight. More direct evidence of atoms came with the erratic jittery motion, known as “Brownian motion”, of pollen grains suspended in water noted by the botanist Robert Brown in 1827 (even if Brown himself never quite solved the problem of their mysterious behavior. But, in order to identify exactly what makes one atom different from another, it was necessary to look inside an atom. In 1895, the British physicist J. J. Thomson had discovered the first sub-atomic particle, the electron, a tiny particle 2,000 smaller than even a hydrogen atom, although he had actually envisaged an atom as a multitude of these tiny negatively-charged electrons embedded in a diffuse ball of positive charge, like raisins in a pudding.
Experiments in 1907 by the New Zealander Ernest Rutherford, however, showed that the vast majority (more than 99.9%) of the mass of an atom was packed into a tiny central nucleus (about 100,000 times smaller than the overall atom, depending on the particular element, sometimes compared to the head of a pin in a football stadium) orbited by the even tinier electrons, like planets around a sun. Coming across the nucleus inside an atom has been likened to finding a pea suspended in the space of a cathedral. So, for the first time, the familiar solid world around us was revealed to be composed of mainly empty space. Later, in 1913, Rutherford’s model of the atom was fleshed out and refined by the Danish physicist Niels Bohr. It was not until as late as 1980, though, with the invention of the Scanning Tunnelling Microscope of Gerd Binnig and Heinrich Rohrer, that the first atoms were actually "seen", showing materials to be composed of spherical atoms stacked row on row, much as Democritus had envisaged. As technology improved, the nucleus of an atom was shown to consist of a combination of positively-charged protons and zero-charged neutrons. By the late 1960s, it was known that protons and neutrons themselves have size and extension and so could not be considered fundamental particles, but were made up of yet smaller matter. Each proton and neutron was revealed to be comprised of three quarks, elementary particles which come in six different flavors (up, down, top, bottom, strangeness and charm) and which are several orders of magnitude smaller than the protons and neutrons they make up. The very names of these properties tell of their arbitrariness, and it is impossible to say what they might look like in the physical world. The name quark was taken from a particularly difficult James Joyce novel by Murray Gell-Man (who was instrumental in their discovery), and was used in preference to Richard Feynman’s more light-hearted suggestion of the “parton” (after Dolly Parton). A hypothetical sub-component of quarks and electrons, dubbed the preon, was postulated in the 1970s and 1980s, but it has since been largely abandoned as superfluous and experimentally unproven. Quarks and electrons do in fact appear to be elementary particles.
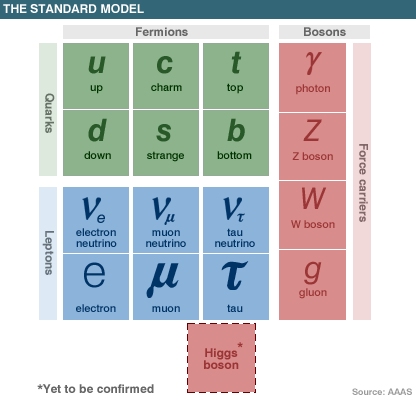
The current Standard Model of particle physics consists of 16 elementary particles: up, down, top, bottom, charm and strange quarks; electrons; muon and tau leptons; three types of neutrinos; Z and W bosons; photons; and gluons. These particles are acted upon by three fundamental forces: the electromagnetic force, the strong nuclear force and the weak nuclear force. In addition to these particles, the Higgs boson (dubbed the "God particle" by the popular media) is an integral part of the model, as it explains why the other elementary particles (with the exception of gluons and photons) have mass, and therefore why matter, in general, has mass. The Standard Model has proved to be a remarkably robust and successful model, and several particles which were merely theoretical when it was first formulated in the mid-1970s have since been observed experimentally, including most recently (2012) the elusive Higgs boson. But, to return to Rutherford’s findings in the early 20th Century, though, it quickly became abundantly clear that his findings were completely at odds with the known physics of the time. According to the classical theory of electromagnetism developed in the second half of the 19th Century by James Clerk Maxwell (who demonstrated that electricity, magnetism and even light are all manifestations of the same phenomenon, the electromagnetic field), any charged particle which accelerates or changes speed or direction gives out electromagnetic waves. In that case, then, an electron circling a nucleus should in theory be continually sapped of its energy, and should therefore spiral into, and collide with, the nucleus of the atom within micro-seconds. So, even before 1910, it was becoming apparent that atoms should not even be able to exist according to classical theory! Enter quantum theory (although some further explanation is still needed before that particular question can be directly addressed).
|
Back to Top of Page
Introduction | Main Topics | Important Dates and Discoveries | Important Scientists | Cosmological Theories | The Universe By Numbers | Glossary of Terms | A few random facts | Blog | Gravitational Lensing Animation | Angular Momentum Calculator | Big Bang Timeline
NASA Apps - iOS | Android
The articles on this site are © 2009-.
If you quote this material please be courteous and provide a link.
Citations | Sources | Privacy Policy